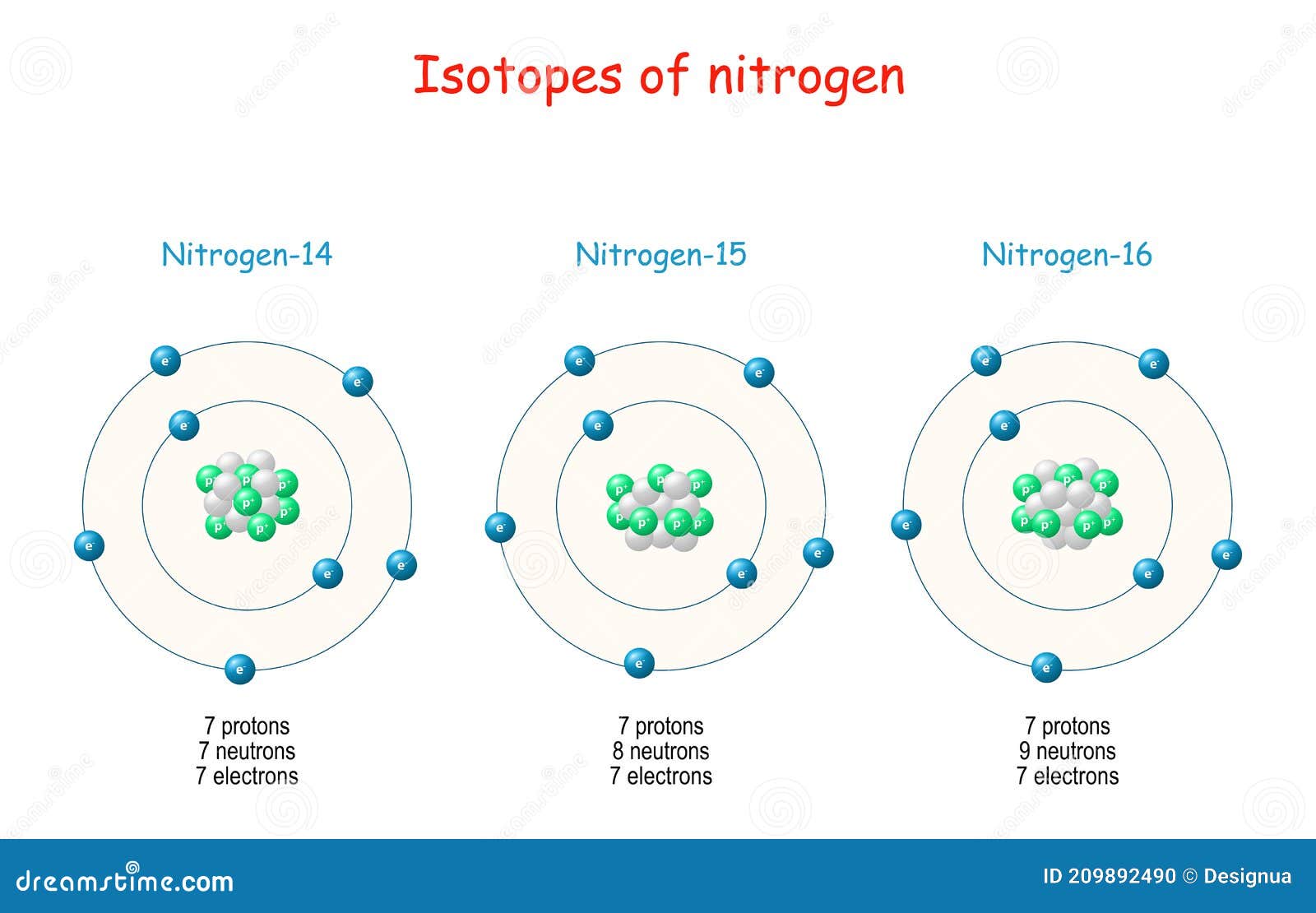
Since the electron in the excited atom is unstable, it returns back to its original level with emission of the same quantum of energy ( emission spectrum ) in the form of radiant light that appears in the form of characteristic spectral line of a certain wavelength and frequency. When the electron acquires a quantity of energy – known a s quantum – by heating or by electric discharge, the electron jumps temporarily to a higher energy level, This is in case that the absorbed quantum of energy is equal to the difference in energies between the two levels, and the atom is known as excited atom. Įach electronin the atom has a definite amount of energy depending on the distance between its energy level and the nucleus, the energyof any level increases as its radius increases, Each energy level expressed by a whole number called the principal quantum number ( n ). Įlectrons orbit the nucleus in definite allowed energy levels, They can not be found at intermediate distances, at which electron moves from an energy level to another one via a complete jumping. Įlectronsorbit the nucleus in a rapid movement without emission or absorption of any amount of energy and the atom in this case named stable atom. During the revolving of the electron around the nucleus, a centrifugal force arises which is equal to the attraction force of the nucleus on the electron.The number of negative electrons ( revolving around the nucleus ) equals the number of positive protons inside the nucleus.A positively charged nucleus exists in the center of the atom.Points that agree with Rutherford’s postulates The study of atomic spectra is considered the key which solved the puzzle of the atomic structure, That was the work of the Danish scientist Niels Bohr upon which he was rewarded the Nobel Prize in 1922. īohr’s atomic theory Bohr’s atomic model ( 1913 ) Īpplication : The line spectrum of hydrogen atom appears ( on examining ) as four coloured lines separated by dark areas, It was found experimentally that the spectral lines are essential characteristics for each element, because there are no two elements have the same spectral lines. Line spectrum is a type of spectrum composed of a small number of restricted coloured lines separated by dark areas, The radiant light is named as line spectrum, because it is composed of a limited number of restricted coloured lines which are separated by dark areas. On examining this radiant light by a device called spectroscope, it was found that it is composed of a limited number of restricted colored lines separated by dark areas, So, it is called line spectrum, It is worth mentioning that the physicists – at that time – were not able to explain this phenomenon. Under these circumstances, no electron could lose energy because no electron could move down to a lower energy level.On heating atoms of a pure elements – in gaseous or vapor state – to a high temperature or exposing them to a low pressure in an electrical discharge tube, they emit a radiation called emission spectrum ( line spectrum ). Bohr worked out rules for the maximum number of electrons that could be in each energy level in his model, and required that an atom in its normal state (ground state) had all electrons in the lowest energy levels available. In this situation, no person could move down because all of the lower rungs are full. If the ladder had five people on it, they would be on the lowest five rungs. Other rules for the ladder are that only one person can be on a rung in the normal state, and the ladder occupants must be on the lowest rung available. Suppose we had such a ladder with 10 rungs. The only way you can be on that ladder is to be on one of the rungs, and the only way you could move up or down would be to move to one of the other rungs. It would be like a ladder that had rungs only at certain heights. The energy levels are quantized, meaning that only specific amounts are possible. 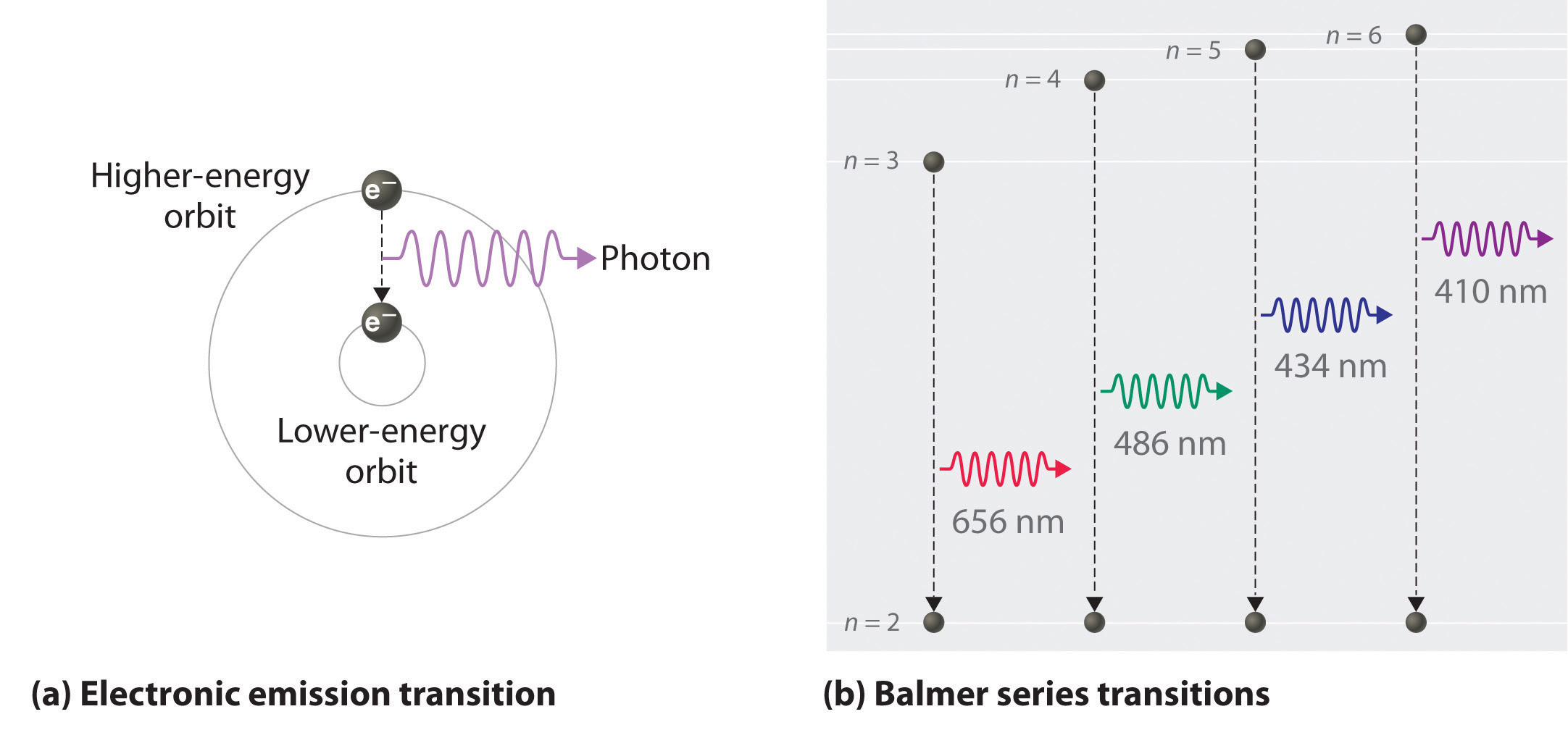 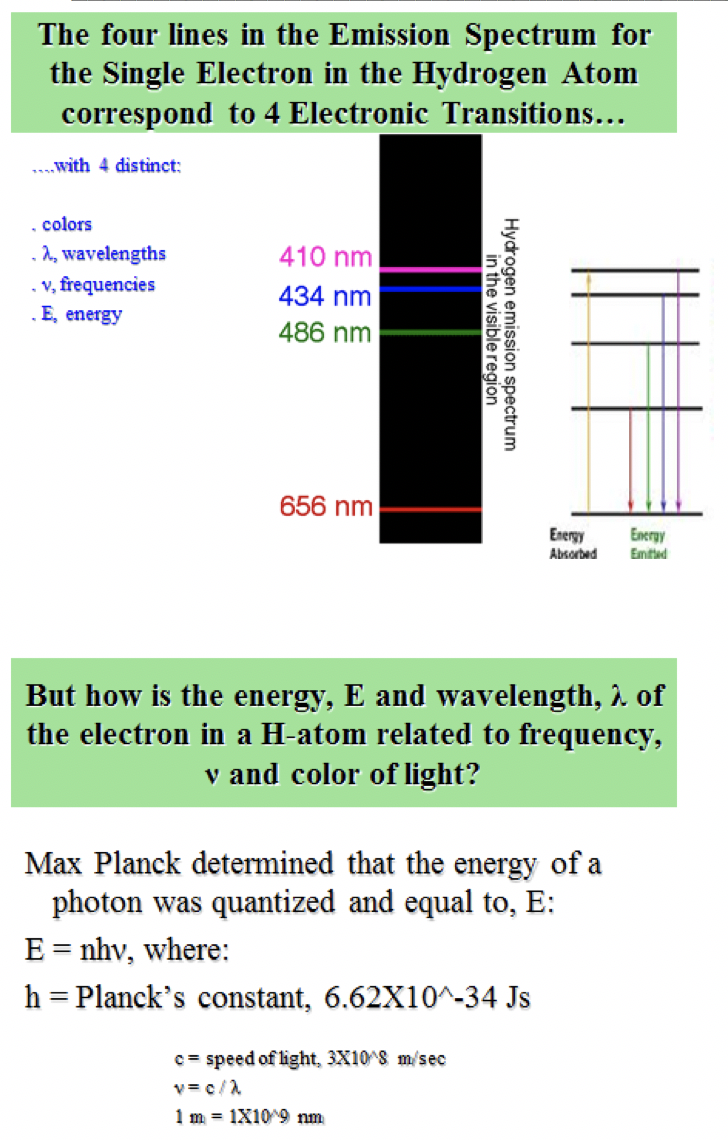 
Note that the spacing between rungs gets smaller at higher energies (CC BY-NC Ümit Kaya)īohr hypothesized that the only way electrons could gain or lose energy would be to move from one energy level to another, thus gaining or losing precise amounts of energy. \): The energy levels of the electrons can be viewed as rungs on a ladder.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
